



🔔Join APTI PLUS Prelims Mirror 2026 | All India Open Mock Test Series on 12th April, 26th April & 3rd May 2026 |Register Now!
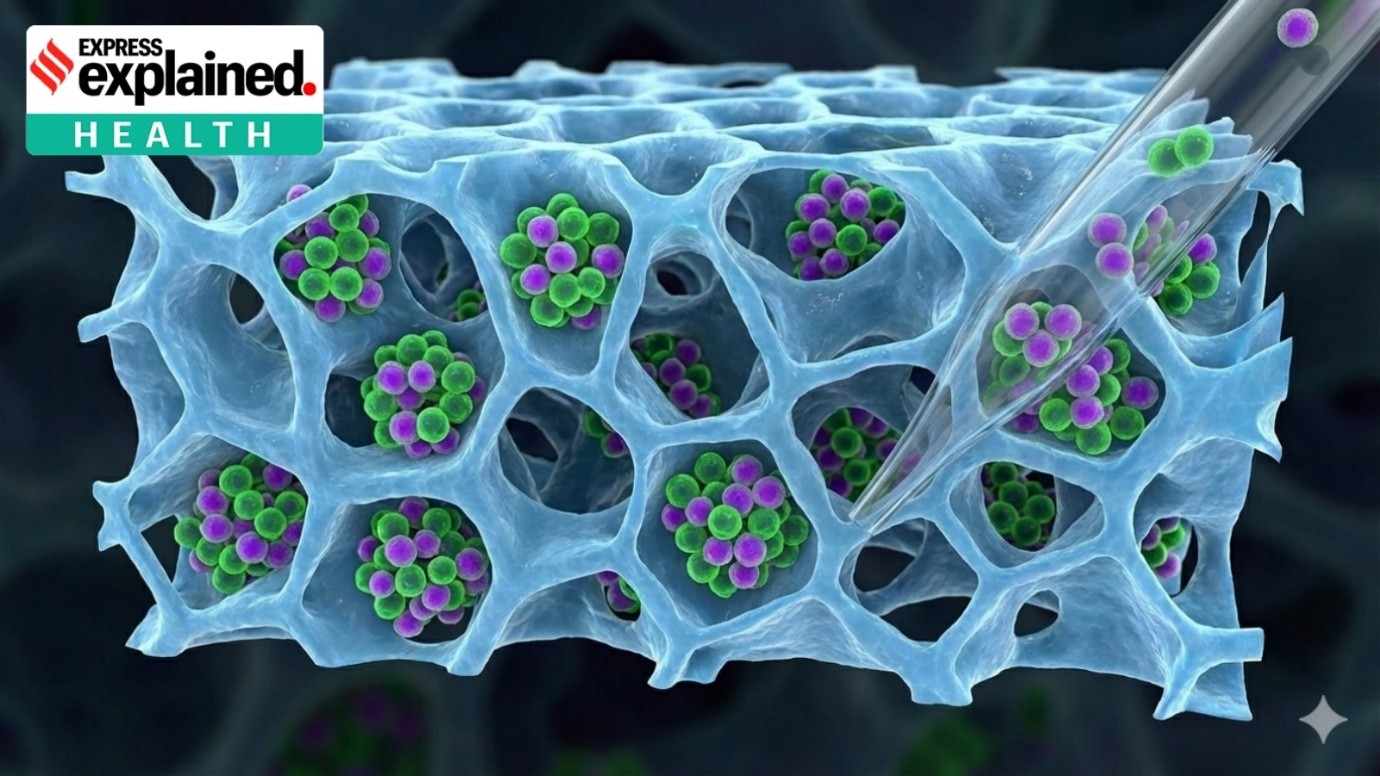
Context: Pune-based Serum Institute of India (SII) unveiled India’s first indigenously developed pneumococcal vaccine.
https://indianexpress.com/article/explained/pneumosil-vaccine-pneumococcal-disease-7124129/






© 2026 iasgyan. All right reserved