Description

Disclaimer: Copyright infringement not intended.
Context: A new study raises concerns about the use of the common herbicide clethodim.
About the study and findings
- It was a collaborative study of researchers from Kasturba Medical College (KMC), Manipal Academy of Higher Education (MAHE), Manipal, and Yenepoya Research Center, Mangaluru.
- The findings were published in the journal Chemosphere.
- The research has unveiled alarming insights into the potential impact of the widely used herbicide clethodim on male reproductive health.
- The study reveals a spectrum of effects including a reduction in testicular weight, a decrease in germ cell population, lower levels of serum testosterone, abnormalities in sperm, and compromised preimplantation embryo development.
Importance of the findings
- These findings necessitate further investigation and reconsideration of the use of such herbicides to ensure the well-being of both humans and our environment.
- There is a necessity for molecular-level screenings to comprehend the potential effects of herbicides on human and environmental health.
- The researchers underlie the urgency of comprehensive testing for this post-emergent herbicide.
- There is a need for revisiting the application of the herbicid
- Advocacy for upgraded standards in the formulation of new agents for similar uses can be done.

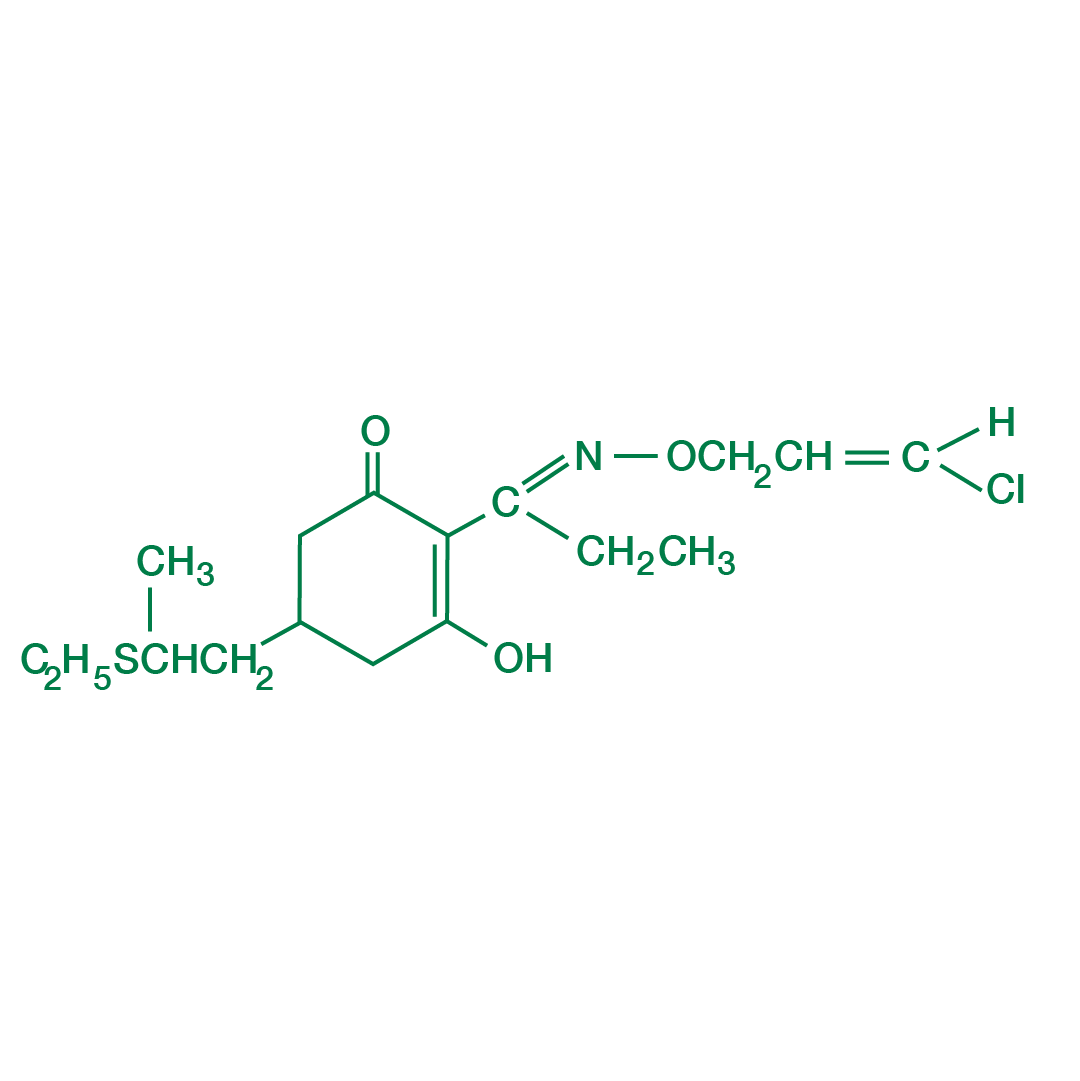
About Clethodim
- It is an organic compound.
- It is a member of the cyclohexanedione family of herbicides.
- It is used to control grasses, especially Lolium rigidum.
- Clethodim has been an approved herbicide in India and globally for some time.
Regulations of herbicides and pesticides in India
Central Insecticides Board and Registration Committee
The import, manufacture, sale, transport, distribution, and use of insecticides and herbicides is regulated by the central insecticide board and registration committee in India.
The pesticide in India is regulated by the following two government bodies
-
- Central Insecticides Board and Registration Committee (CIBRC) and
- Food Safety and Standards Authority of India (FSSAI)
Central Insecticide Board (CIB)
- The Central Insecticides Board advises the central government and state governments on technical matters arising out of the administration of this act and to carry out the other functions assigned to the board by or under this act.
- The CIB committee regulates the entry of new active substances or formulations of pesticides in India.
- It is the apex body involving the various experts that evaluate the registrability of a particular molecule in India.
- After the approval from CIB the new molecule is included in the Insecticide Schedule.
- Once CIB approves the new molecule inclusion, the applicant can start the registration process.
Registration Committee (RC)
- Once the new molecule is included in the insecticide schedule, the applicant is required to obtain the registration of the new molecule under various 9(3) categories to legally sell, trade, manufacture or import the new molecule or its formulation in India.
- The RC facilitates the registration process through an online portal. The various experts in the RC scrutinize the formulae and verify claims made by the importer or the manufacturer as regards their efficacy and safety to human beings and animals.
- After the requirements are met satisfactorily, RC presents the Certificate of Registration (CR) to the applicant.
Food Safety and Standards Authority of India (FSSAI)
- As per the Food Safety and Standards Act, 2006, the FSSAI has constituted a Scientific Panel with relevant experts which recommends Maximum Residue Level (MRL).
- In India, the State Agricultural Universities (SAUs)/Indian Council of Agricultural Research (ICAR) generate the multi-location supervised field trial data for pesticide and herbicide residues following GAP on registered crops approved by CIB&RC.
- The Food Safety and Standard Authority of India (FSSAI) under the Ministry of Health and Family Welfare evaluates the supervised trial residue data based on the approved GAP for fixation of MRL, keeping in view the dietary exposure and risk assessment of the pesticide.
|
PRACTICE QUESTION
Q. Recent findings on adverse health impacts of pesticides and insecticides in India highlight the lacunae in their quality assessment, risks and the overall insecticide governance mechanism in India. Critically evaluate. ( 250 words)
|

https://www.thehindu.com/sci-tech/health/a-silent-threat-to-male-reproductive-health-new-study-raises-concerns-on-use-of-common-herbicide-clethodim/article67230478.