



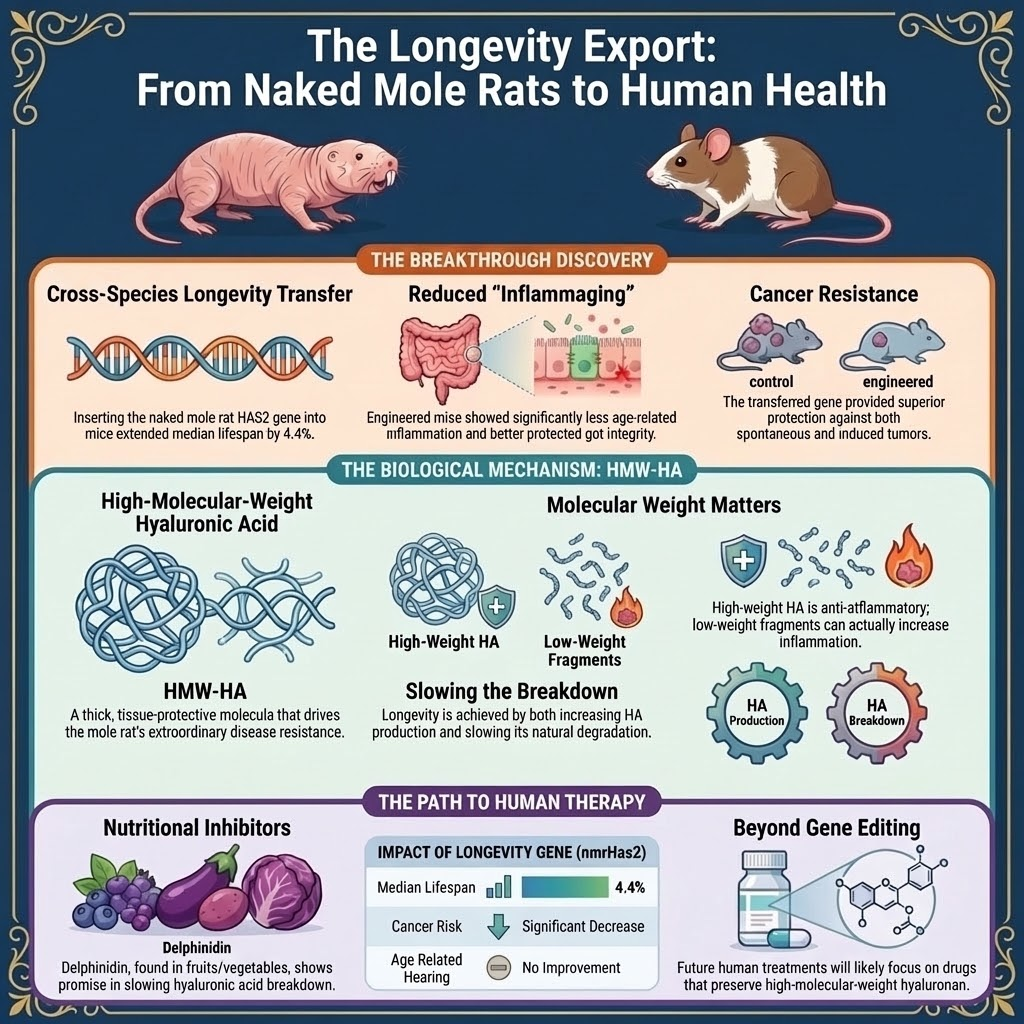
Researchers transferred the naked mole rat longevity gene HAS2 to mice, increasing high-molecular-weight hyaluronic acid that reduces cancer risk and inflammaging. The mice showed better healthspan and slightly longer lives. The discovery may enable human anti-aging therapies but raises ethical, safety, and equity concerns.

Copyright infringement not intended
Picture Courtesy: SCITECHDAILY
Context
Scientists transferred a longevity gene from the mole rat to mice, which led to improved health and a slightly longer lifespan in the mice, demonstrating that longevity mechanisms can be shared across species
What is Longevity Gene Transfer?
Longevity gene transfer refers to a variety of experimental gene therapies designed to extend lifespan and improve healthspan by introducing or enhancing specific "longevity genes."
Adeno-Associated Virus (AAV) vectors are primarily used in research to deliver genes into cells. These "non-disease-causing" vectors offer long-lasting therapeutic protein expression.
Recent Breakthroughs in Gene Transfer
Species-to-Species Transfer (Naked Mole Rat to Mice): Rochester University researchers successfully transferred the naked mole rat's hyaluronan synthase 2 (HAS2) gene into mice.
Supercentenarian Gene for Heart Health: Scientists have identified a gene variant called LAV-BPIFB4, found in individuals who live past 100.
Telomerase (TERT) Therapy: Delivering the TERT gene (which maintains the protective caps on chromosomes called telomeres) via viral vectors has extended mouse lifespan by up to 24% without increasing cancer risk.
Limitations and Ethical Considerations
Scientific Nuances: The lifespan extension in mice was modest, and the gene transfer did not prevent all age-related ailments like hearing loss.
Equity and Access: A primary ethical concern is the risk of a "longevity divide," where expensive therapies could exacerbate social inequalities.
Socio-Economic Impact: Extending human lifespan could strain retirement systems, employment, and global resource allocation.
Medicalizing Aging: There is a risk of treating natural aging as a "disease," which could alter the fundamental human experience of life and mortality.
Way Forward
The successful transfer of a longevity gene is a monumental step that shifts the scientific focus from merely extending lifespan to enhancing healthspan—adding quality life to years, not just years to life.
Source: SCITECHDAILY
|
PRACTICE QUESTION Q. With reference to the recent research on longevity, what is High Molecular Weight Hyaluronic Acid (HMW-HA) primarily associated with? (a) Triggering inflammation and tumour progression. (b) Anti-inflammatory properties and cancer resistance. (c) Increasing the rate of cellular aging. (d) Being a synthetic molecule developed in laboratories. Answer: B Explanation: Recent longevity research has identified High Molecular Weight Hyaluronic Acid (HMW-HA) as a critical factor in their extraordinary lifespan and immunity to cancer. |
The 'longevity gene' is the naked mole rat's version of the hyaluronan synthase 2 (HAS2) gene. It is uniquely adapted to produce large quantities of High Molecular Weight Hyaluronic Acid (HMW-HA), which is linked to longevity and disease resistance.
HMW-HA has strong anti-inflammatory and tissue-protective properties. It helps combat 'inflammaging' (chronic, low-grade inflammation associated with aging) and has been shown to provide remarkable resistance against cancer by preventing cancerous cells from multiplying.
Directly transferring genes to humans is not the immediate goal. Instead, the research suggests two therapeutic pathways: 1) Developing drugs (like delphinidin) that slow the breakdown of the body's existing HMW-HA, and 2) Creating therapies that encourage our own cells to produce more of this beneficial molecule.

© 2026 iasgyan. All right reserved