




Disclaimer: Copyright infringement not intended.
Context
Details
Factory Setup and Production Plans
Conversion of Petroleum Vehicles into Electric Vehicles
Challenges and Regulatory Environment
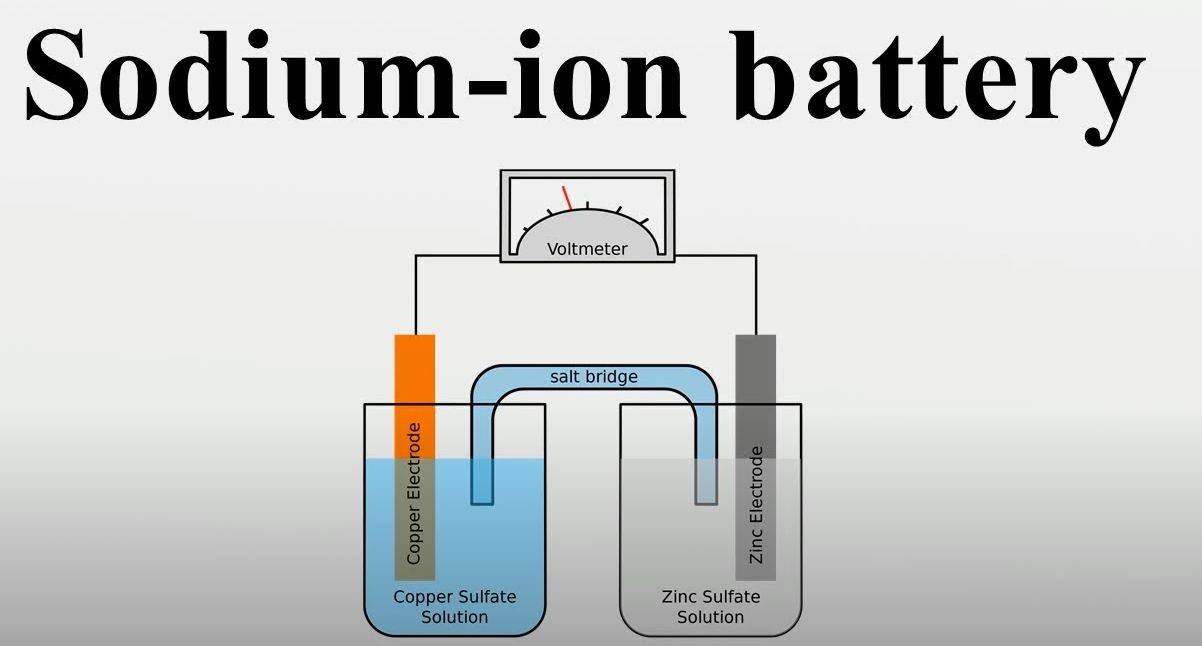
Introduction to Sodium Ion Batteries
Battery Landscape and Evolution
Role of Energy Storage Technologies
Need for Sodium Ion Batteries
Basic Principles and Components
Electrolyte: Facilitator of Ion Movement
Anode Materials: Sodium Storage Mechanisms
Cathode Materials: Reversible Sodium Intercalation
Working Principle: Charge and Discharge Processes
Comparison with Other Battery Technologies
Sodium Ion vs. Lithium-ion Batteries
Resource Abundance and Cost Factors
Energy Density and Power Density Comparison
Safety Considerations
Pros and Cons of Sodium Ion Batteries
Pros
Cons
Anode Materials
Carbon-based Materials
Hard Carbon
Hard carbon materials, derived from sources like graphite and biomass, offer good cycling stability and reversible sodium intercalation. However, they may suffer from lower initial capacity compared to other materials.
Soft Carbon
Soft carbon materials, often obtained from petroleum residues, provide higher initial capacity but might experience capacity fading over cycling due to structural changes.
Transition Metal Oxides
Transition metal oxides, such as tin-based compounds, offer high theoretical capacity for sodium storage. However, they can experience significant volume changes during cycling, leading to electrode degradation.
Phosphates and Silicates
Phosphate and silicate-based materials provide good cycling stability and reversible reactions, making them potential candidates for long-lasting sodium ion batteries.
Cathode Materials
Prussian Blue Analogs
Prussian blue analogs, such as iron hexacyanoferrates, are promising cathode materials for sodium ion batteries. They offer a reversible sodium insertion/extraction mechanism, high capacity, and good cycling stability. Their open framework structure allows for the accommodation of sodium ions during charging and discharging.
Polyanionic Compounds
Polyanionic cathode materials, like sodium iron phosphates, exhibit stable cycling performance due to their robust crystal structures. They enable reversible sodium intercalation while maintaining structural integrity, making them suitable for long-term battery operation.
Transition Metal Chalcogenides
Transition metal chalcogenides, including sulfides and selenides, provide high theoretical capacities and favorable electronic conductivity. However, their large volume changes during cycling can lead to mechanical stress and performance degradation over time.
Electrolytes and Ionic Conductors
Liquid Electrolytes
Liquid electrolytes, typically composed of sodium salts dissolved in organic solvents, facilitate ion transport between the anode and cathode. While they offer high ionic conductivity, they also pose safety risks due to flammability and potential leakage.
Solid Electrolytes
Solid electrolytes offer improved safety and stability compared to liquid counterparts. They can suppress dendrite formation and eliminate the risk of electrolyte leakage. However, challenges remain in achieving high ionic conductivity and interface compatibility with electrode materials.
Importance of Ion Mobility
Ionic conductivity is crucial for efficient charge/discharge processes. Enhancing ion mobility in both the electrolyte and electrode materials is essential to achieve high-performance sodium ion batteries.
Cell Chemistry and Reactions
Sodium Insertion/Extraction Mechanisms
Sodium ion batteries involve reversible insertion/extraction of sodium ions into/from the electrode materials during charging and discharging. Understanding the mechanisms of these electrochemical reactions is key to optimizing battery performance and cycle life.
Intercalation vs. Conversion Reactions
Different materials exhibit varying reaction mechanisms. Some materials undergo intercalation reactions, where sodium ions fit into the crystal lattice of the electrode material. Others undergo conversion reactions, where chemical transformations occur between the electrode and sodium ions.
Battery Performance and Metrics
Energy Density and Specific Energy
Energy density refers to the amount of energy stored per unit volume or mass of the battery. While sodium ion batteries generally have lower energy density than lithium-ion batteries, they can still offer sufficient energy density for certain applications.
Power Density and Specific Power
Power density measures how quickly a battery can deliver energy. Sodium ion batteries can provide competitive power density, enabling rapid energy delivery for applications requiring high power outputs.
Cycle Life and Calendar Life
Cycle life refers to the number of charge/discharge cycles a battery can undergo while maintaining a specified capacity. Calendar life refers to the battery's operational lifespan even when not in use. Optimizing both aspects is crucial for the economic viability of sodium ion batteries.
Challenges and Solutions
Crystallographic Changes in Electrode Materials
Some electrode materials undergo structural changes during sodium ion insertion/extraction, leading to capacity fading and reduced performance. Designing materials with stable crystal structures and accommodating volume changes is a research focus.
Sodium Dendrite Formation
Similar to lithium-ion batteries, sodium ion batteries can suffer from dendrite growth, which can short-circuit the battery and lead to safety hazards. Strategies such as electrolyte additives and solid electrolytes are being explored to mitigate this issue.
High Operating Temperature
Some sodium ion batteries may require elevated temperatures to achieve desirable performance. Developing materials and designs that operate efficiently at ambient temperatures is crucial for practical applications.
Capacity Fading Over Cycles
Repeated cycling can lead to capacity degradation due to electrode material fatigue and electrolyte breakdown. Improving electrode stability and electrolyte design are essential for maintaining consistent performance over the battery's lifespan.
Applications and Market Potential
Grid-Scale Energy Storage
Sodium ion batteries hold potential for large-scale energy storage, helping to stabilize power grids by storing excess energy during low-demand periods and releasing it during peak demand. Their scalability and potentially lower cost make them an attractive option for grid operators.
Renewable Energy Integration
Sodium ion batteries can facilitate the integration of intermittent renewable energy sources, such as solar and wind, by storing surplus energy and delivering it when energy generation is low. This enhances grid stability and reduces reliance on fossil fuels.
Electric Vehicles and Portable Electronics
While their energy density might be lower than lithium-ion batteries, sodium ion batteries can still find applications in electric vehicles (EVs) and portable electronics. They offer a more sustainable alternative to lithium-ion technology and can help reduce the environmental impact of battery production.
Research and Development Trends
Advancements in Electrode Materials
Ongoing research focuses on discovering new electrode materials with improved capacity, stability, and safety. Nanomaterials, composites, and hybrid structures are being explored to enhance performance.
Exploration of New Electrolytes
The development of solid-state electrolytes with high ionic conductivity is a major research direction. These electrolytes can offer enhanced safety, wider operating temperature ranges, and compatibility with various electrode materials.
Emerging Technologies and Innovations
Nanotechnology, 3D printing, and advanced manufacturing techniques are being employed to optimize battery design and enhance performance. Innovations in battery management systems and predictive modeling are also contributing to the advancement of sodium ion battery technology.
Environmental and Safety Considerations
Sodium Abundance and Sustainability
The abundance of sodium in Earth's crust makes sodium ion batteries a potentially sustainable energy storage solution. This contrasts with lithium-ion batteries, which rely on limited lithium resources.
Toxicity and Environmental Impact
Sodium ion batteries generally have lower toxicity concerns compared to lithium-ion batteries. However, environmental considerations regarding the extraction of raw materials, manufacturing processes, and end-of-life disposal remain important.
Safety Measures and Thermal Stability
Solid electrolytes and improved electrode materials can enhance the safety of sodium ion batteries by reducing the risk of dendrite formation, thermal runaway, and electrolyte leakage.
Commercialization and Industry Players
Companies Developing Sodium Ion Battery Technology
Several companies are actively engaged in the research, development, and commercialization of sodium ion battery technology. Collaborations between startups, established battery manufacturers, and research institutions are driving innovation in this field.
Current Market Status and Future Projections
While sodium ion batteries are still in the early stages of commercialization compared to lithium-ion batteries, the market is expected to grow as technology advancements continue. As the demand for large-scale energy storage and sustainable energy solutions increases, sodium ion batteries are poised to play a significant role.
Regulations and Standards
Battery Safety Standards
As sodium ion battery technology progresses, safety standards are being established to ensure the safe manufacturing, transportation, and usage of these batteries. These standards help mitigate risks and ensure consistency in quality and safety.
Regulatory Frameworks for Sodium Ion Batteries
Regulatory bodies and governments are gradually developing frameworks to govern the production, use, and disposal of sodium ion batteries. These frameworks aim to address environmental concerns, ensure safety, and promote responsible battery development
Conclusion
Sodium ion batteries represent a promising alternative to existing battery technologies, offering advantages in terms of resource availability, safety, and potential cost reduction. As research and development efforts continue, sodium ion batteries are expected to become increasingly competitive in various applications, from grid-scale energy storage to portable electronics and electric vehicles. Their potential to address both energy and environmental challenges makes them a key player in the transition to a more sustainable energy future.
|
PRACTICE QUESTION Q. Discuss the significance and potential applications of sodium-ion batteries in the context of India's energy transition. What are the key advantages and challenges associated with sodium-ion battery technology, and how can the government and industry collaborate to promote its adoption and development? (250 Words) |






© 2026 iasgyan. All right reserved