




Copyright infringement not intended
Picture Courtesy: timesofindia.indiatimes.com
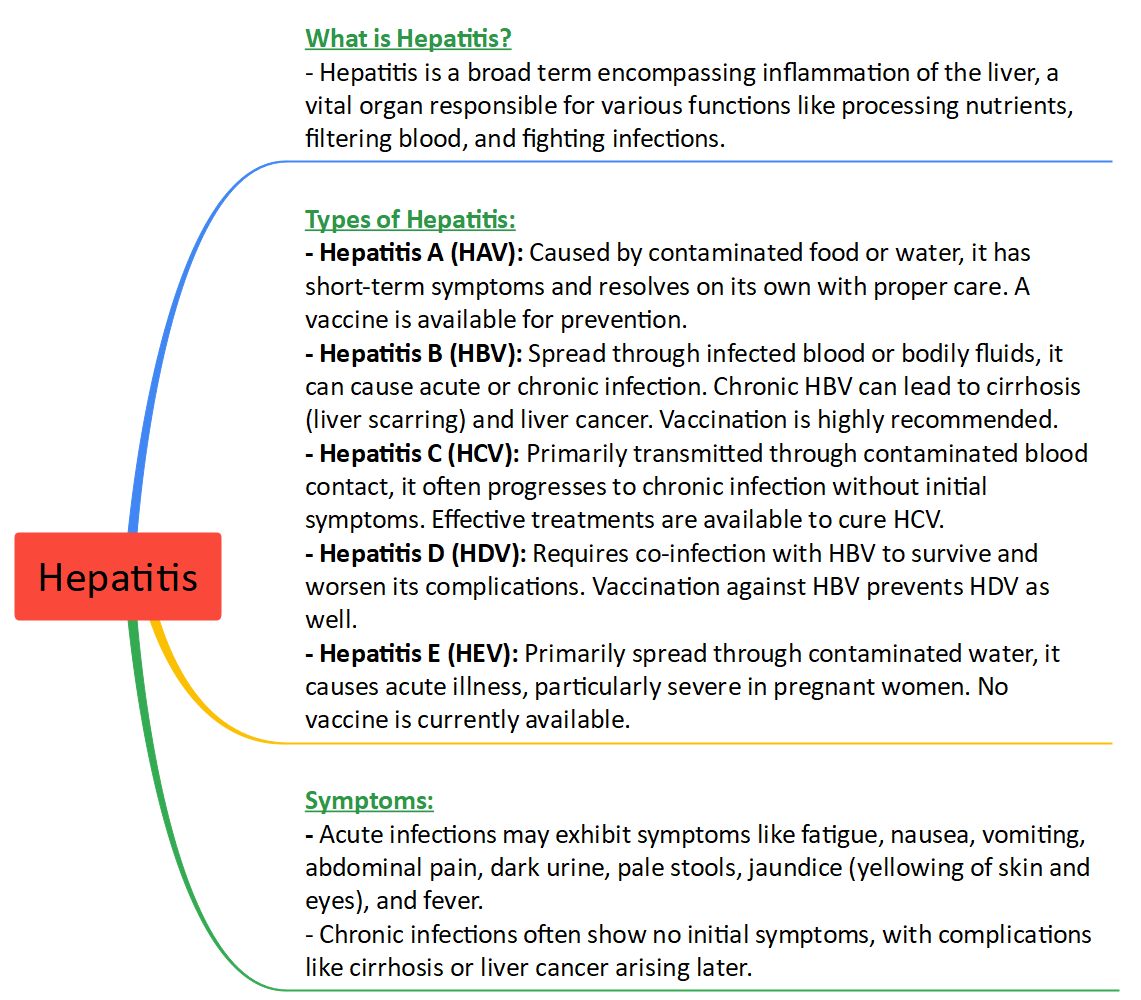
Context: Indian Immunologicals Ltd (IIL), a subsidiary of the National Dairy Development Board (NDDB) and a leading biopharmaceutical company in Hyderabad, launched India's first indigenously developed Hepatitis A vaccine named 'Havisure.'
India's First Hepatitis A Vaccine 'Havisure' Launch
Must Read Articles:
HEPATITIS C: https://www.iasgyan.in/daily-current-affairs/hepatitis-c
|
PRACTICE QUESTION Q. What is the significance of “Havisure”, frequently seen in the news: A) It requires a unique three-dose schedule for complete protection. B) It offers significantly longer-lasting immunity compared to existing vaccines. C) It represents a major step towards self-reliance in vaccine production for India. D) It is significantly cheaper than other available Hepatitis A vaccines. Answer: C Explanation: Havisure's development marks a significant milestone in India's efforts to become self-sufficient in vaccine production, reducing reliance on imports and potentially making the vaccine more accessible to the population. |






© 2026 iasgyan. All right reserved