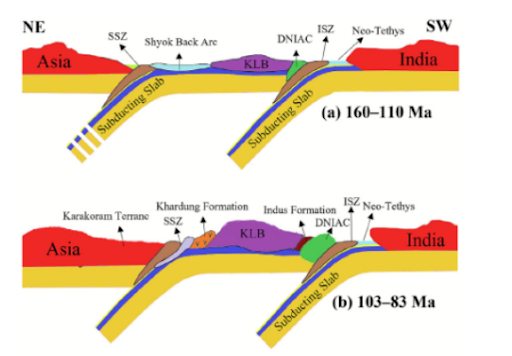
Description
Context: India's largest ever trial has found convalescent plasma therapy ineffective in Covid-19.
- Recently published findings on convalescent plasma therapy on Covid-19 patients have triggered a debate over its efficacy.
- After the country’s largest such trial, known by the acronym PLACID, found that convalescent plasma was ineffective in arresting Covid-19, the Indian Council of Medical Research (ICMR) has been considering dropping this option from the national guidelines.
What is plasma therapy?
- Plasma is the liquid part of the blood.
- Convalescent plasma, extracted from the blood of patients recovering from an infection, is a source of antibodies against the infection.
- The therapy involves using their plasma to help others recover.
- For Covid-19, this has been one of the treatment options. The donor would have to be a documented case of Covid-19 and healthy for 28 days since the last symptoms.
What has happened to spark the debate?
- An ICMR study has found convalescent plasma was not associated with a reduction in progression to severe Covid-19 or all-cause mortality.
What are the trial findings?
- While use of convalescent plasma seemed to improve resolution of shortness of breath and fatigue in patients with moderate Covid-19, this did not translate into a reduction in 28-day mortality or progression to severe disease.
- PLACID was a randomised controlled trial in 39 hospitals spread across 14 states and Union Territories and representing 25 cities.
- Progression to severe disease or death at 28 days after enrolment occurred in 44 (19%) of the participants in the intervention arm as compared to 41 (18%) in the control arm .
- A higher proportion of patients in the intervention arm showed resolution of shortness of breath and fatigue at day 7, whereas resolution of fever and cough did not differ between the two arm.
Have there been such results elsewhere?
- In China, a controlled trial of 103 patients with severe Covid-19 reported no effect of convalescent plasma treatment in terms of time to clinical improvement.
- In the Netherlands, the COVID trial, prematurely terminated after 86 patients had been enrolled, could not find any effect on mortality at 60 days.
So, who all are still pushing convalescent plasma therapy?
- The convalescent plasma played a big part in his recovery and many lives have been saved.
- In Maharashtra, the government has been conducting the Platina trial in severely ill patients.
- It is the brainchild of the Medical Education Department and CM Uddhav Thackeray.
What happens if ICMR does remove the therapy from its guidelines?
- The ICMR has been cautious because of the trial findings.
- Guidelines are not necessarily binding and it is too early to dismiss convalescent plasma therapy.
- The authorisation of convalescent plasma as treatment for Covid-19 in India has led to questionable practices such as calls for donors on social media, and the sale of convalescent plasma on the black market.
- Although convalescent plasma is a safe form of treatment when transfused in accordance to the regulations, it involves resource-intensive processes such as plasmapheresis, plasma storage, and measurement of neutralising antibodies.
- A limited number of institutes in India have the capacity to undertake these procedures in a quality-assured manner.
What is the way forward, then?
- This is a new virus, and around the world evidence is still emerging on the best therapeutic options. For example,
- Remdesivir has been sanctioned as a drug of choice by the US drug regulator
- World Health Organization’s Solidarity Trial has found it had little or no effect on 28-day Covid mortality.
- Expert said that use of convalescent therapy has saved some lives but concerns have been raised by the PLACID trial.
- “Covid care is individualised care. Use of the right drugs in the right patient does work.
“The potential harms of the non-immune components of convalescent plasma should be rigorously investigated, only donor plasma with detectable titers of neutralizing antibodies should be given to trial participants, to ensure that the potential for benefit exists for all intervention arm patients.”
https://indianexpress.com/article/explained/the-plasma-therapy-debate-6888258/