Description

Copyright infringement not intended
Picture Courtesy: www.prepladder.com
Context: The United Kingdom’s drug regulator approves the world's first gene therapy treatment for sickle cell disease and thalassemia.
About Casgevy
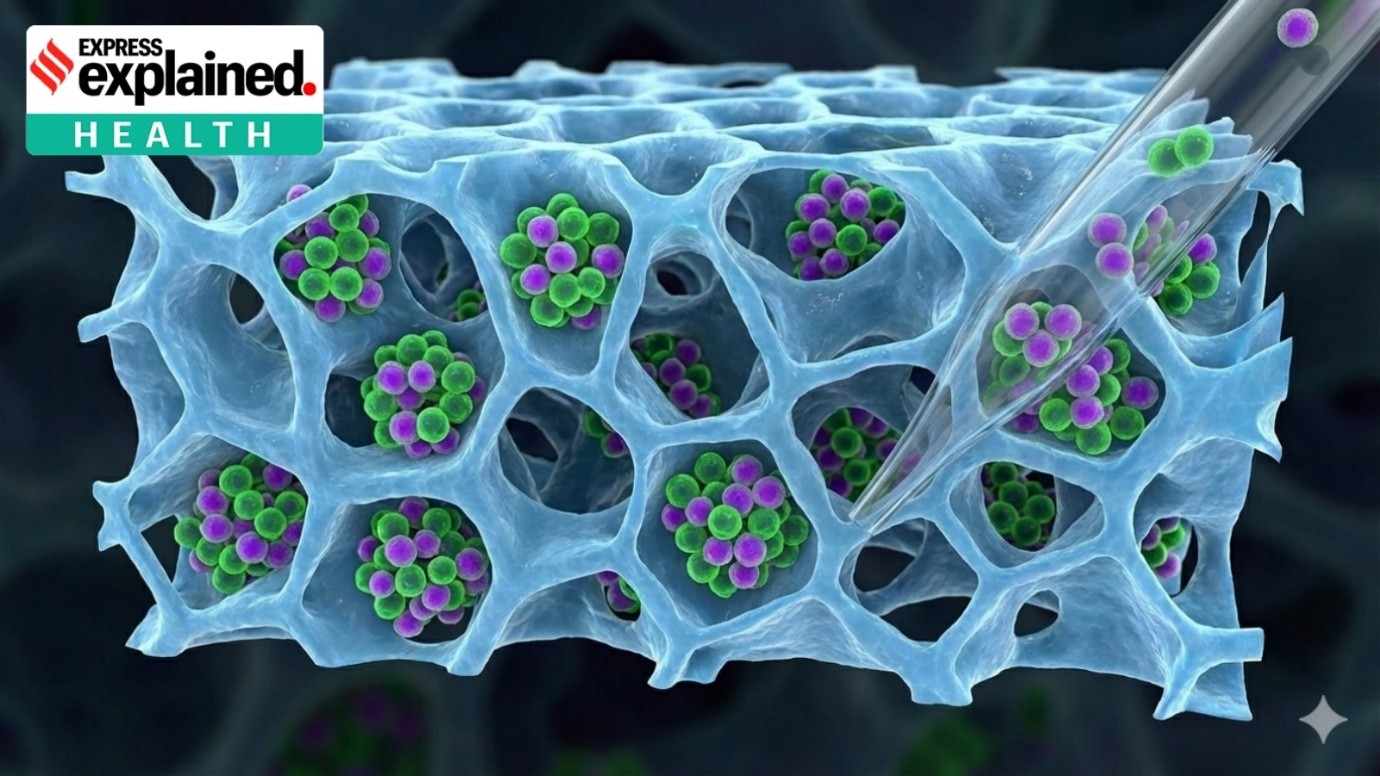
- Casgevy by Vertex Pharmaceuticals (Europe) Ltd. and CRISPR Therapeutics gets approval for patients 12 years old and over with sickle cell disease and thalassemia.
- Both sickle cell disease and thalassemia stem from gene errors affecting haemoglobin, the oxygen-carrying protein in red blood cells.
- Sickle cell disease is prevalent in regions with a history of malaria, like Africa and India, and is more common in certain ethnic groups, such as people of African, Middle Eastern, and Indian descent.
- Previously, bone marrow transplants were the primary long-term treatment, despite their harsh side effects. But with this approval, Casgevy offers hope to thousands affected by these diseases in the UK.
- The decision to greenlight this gene therapy was based on studies involving 29 sickle cell patients, where 28 reported no severe pain problems for at least a year post-treatment. In thalassemia, among 42 patients who received the therapy, 39 did not require a red blood cell transfusion for a year after treatment.
- Casgevy functions by targeting the defective gene in a patient's bone marrow stem cells, allowing the body to produce properly functioning haemoglobin.

Must Read Articles:
Sickle Cell Disease: https://www.iasgyan.in/daily-current-affairs/sickle-cell-disease-24
World Thalasemmia Day: https://www.iasgyan.in/daily-current-affairs/world-thalasemmia-day
|
PRACTICE QUESTION
Q. What is the primary purpose of Casgevy, the recently approved medication?
A. Treatment of hypertension
B. Management of diabetes
C. Gene therapy for sickle cell disease and thalassemia
D. Immunization for viral diseases
Answer: C
Explanation: Casgevy is a gene therapy designed specifically for sickle cell disease and thalassemia.
|