BASICS OF STEM CELLS
In recent years, stem cell therapy has become a very promising and advanced scientific research topic. The development of treatment methods has evoked great expectations. This blog caters to different basic aspects of stem cells.
STEM CELLS CLASSIFICATION
- Stem cells are unspecialized cells of the human body. They are able to differentiate into any cell of an organism and have the ability of self-renewal.
- Stem cells exist both in embryos and adult cells.
- There are several steps of specialization.
- Developmental potency is reduced with each step, which means that a unipotent stem cell is not able to differentiate into as many types of cells as a pluripotent one.
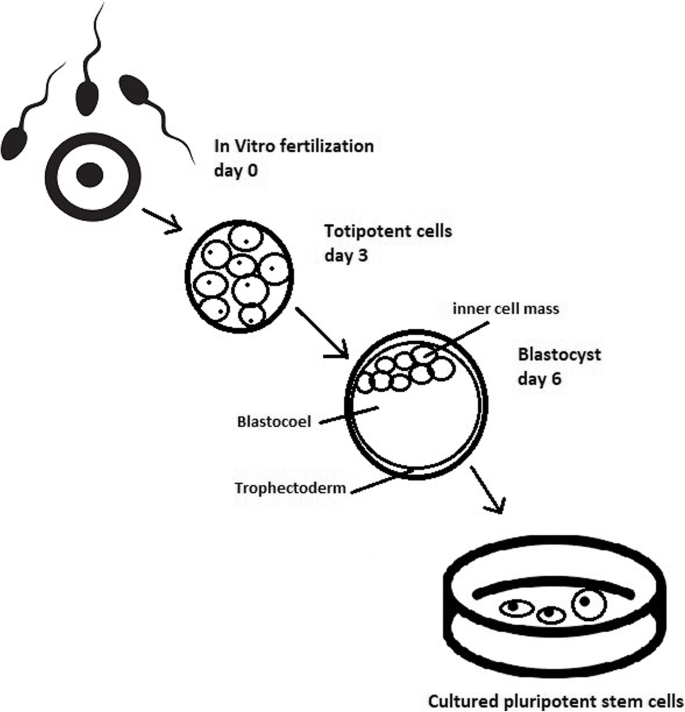
- Totipotent stem cells are able to divide and differentiate into cells of the whole organism. Totipotency has the highest differentiation potential and allows cells to form both embryo and extra-embryonic structures. One example of a totipotent cell is a zygote, which is formed after a sperm fertilizes an egg. These cells can later develop either into any of the three germ layers or form a placenta. After approximately 4 days, the blastocyst’s inner cell mass becomes pluripotent. This structure is the source of pluripotent cells.
- Pluripotent stem cells (PSCs) form cells of all germ layers but not extraembryonic structures, such as the placenta. Embryonic stem cells (ESCs) are an example. ESCs are derived from the inner cell mass of preimplantation embryos. Another example is induced pluripotent stem cells (iPSCs) derived from the epiblast layer of implanted embryos. Their pluripotency is a continuum, starting from completely pluripotent cells such as ESCs and iPSCs and ending on representatives with less potency—multi-, oligo- or unipotent cells. One of the methods to assess their activity and spectrum is the teratoma formation assay. iPSCs are artificially generated from somatic cells, and they function similarly to PSCs. Their culturing and utilization are very promising for present and future regenerative medicine.
- Multipotent stem cells have a narrower spectrum of differentiation than PSCs, but they can specialize in discrete cells of specific cell lineages. One example is a haematopoietic stem cell, which can develop into several types of blood cells. After differentiation, a haematopoietic stem cell becomes an oligopotent cell. Its differentiation abilities are then restricted to cells of its lineage. However, some multipotent cells are capable of conversion into unrelated cell types, which suggests naming them pluripotent cells
- Oligopotent stem cells can differentiate into several cell types. A myeloid stem cell is an example that can divide into white blood cells but not red blood cells.
- Unipotent stem cells are characterized by the narrowest differentiation capabilities and a special property of dividing repeatedly. Their latter feature makes them a promising candidate for therapeutic use in regenerative medicine. These cells are only able to form one cell type, e.g. dermatocytes.
STEM CELL BIOLOGY
- A blastocyst is formed after the fusion of sperm and ovum fertilization.
- Its inner wall is lined with short-lived stem cells, namely, embryonic stem cells.
- Blastocysts are composed of two distinct cell types: the inner cell mass (ICM), which develops into epiblasts and induces the development of a foetus, and the trophectoderm (TE).
- The TE continues to develop and forms the extraembryonic support structures needed for the successful origin of the embryo, such as the placenta.
- As the TE begins to form a specialized support structure, the ICM cells remain undifferentiated, fully pluripotent and proliferative.
- The pluripotency of stem cells allows them to form any cell of the organism. Human embryonic stem cells (hESCs) are derived from the ICM.
- During the process of embryogenesis, cells form aggregations called germ layers: endoderm, mesoderm and ectoderm, each eventually giving rise to differentiated cells and tissues of the foetus and, later on, the adult organism.
- After hESCs differentiate into one of the germ layers, they become multipotent stem cells, whose potency is limited to only the cells of the germ layer. This process is short in human development.
- After that, pluripotent stem cells occur all over the organism as undifferentiated cells, and their key abilities are proliferation by the formation of the next generation of stem cells and differentiation into specialized cells under certain physiological conditions.
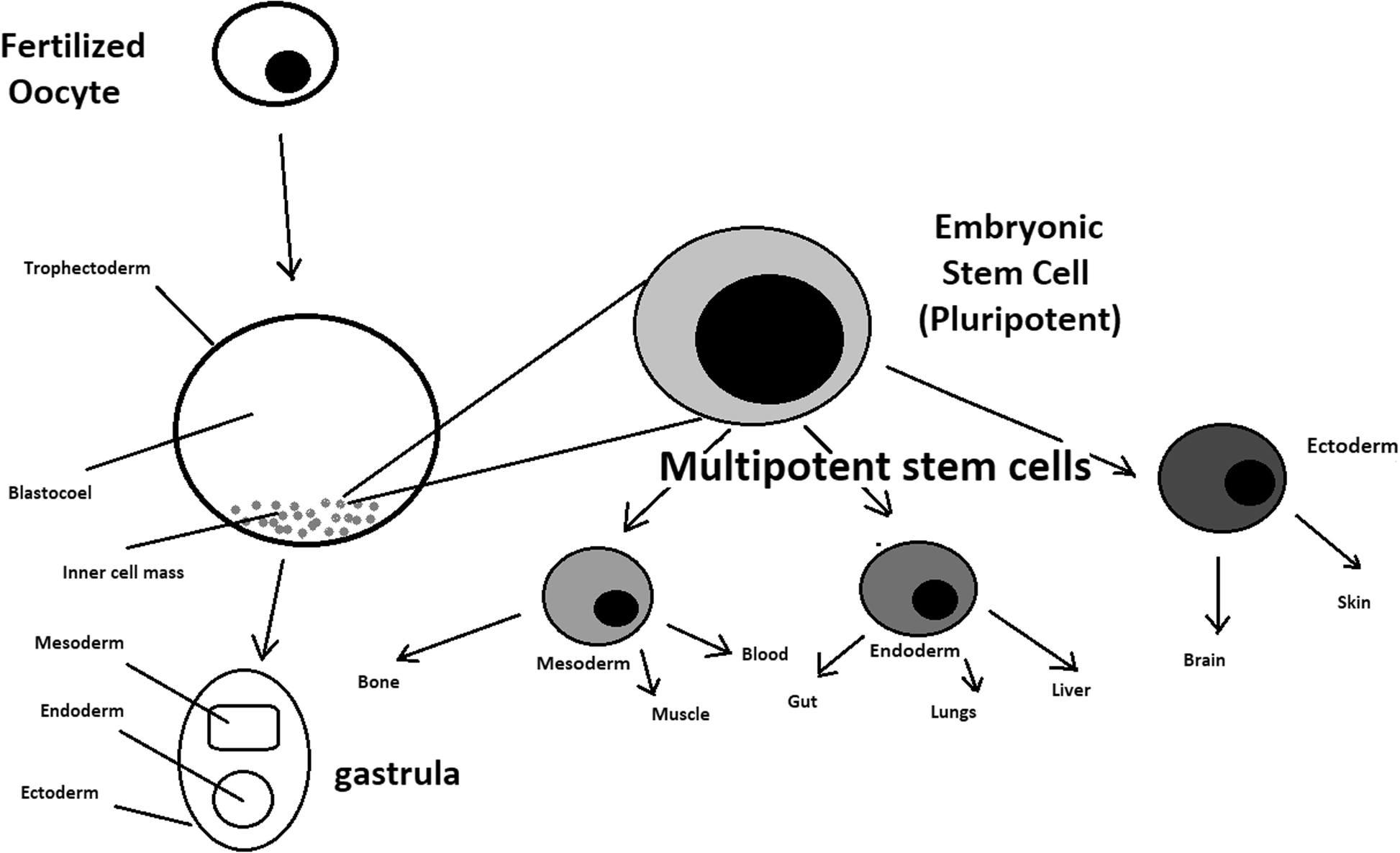
- Signals that influence the stem cell specialization process can be divided into external, such as physical contact between cells or chemical secretion by surrounding tissue, and internal, which are signals controlled by genes in DNA.
- Stem cells also act as internal repair systems of the body.
- The replenishment and formation of new cells are unlimited as long as an organism is alive.
- Stem cell activity depends on the organ in which they are in; for example, in bone marrow, their division is constant, although in organs such as the pancreas, division only occurs under special physiological conditions.
STEM CELL FUNCTIONAL DIVISION
- ESCs are derived from the inner cell mass of the blastocyst, which is a stage of pre-implantation embryo (4 days after fertilization).
- After that, these cells are placed in a culture dish filled with culture medium.
- These cells can be described as pluripotent because they are able to eventually differentiate into every cell type in the organism.
- Somatic or adult stem cells are undifferentiated and found among differentiated cells in the whole body after development. The function of these cells is to enable the healing, growth, and replacement of cells that are lost each day.
- These cells have a restricted range of differentiation options. Among many types, there are the following:
- Mesenchymal stem cells are present in many tissues. In bone marrow, these cells differentiate mainly into the bone, cartilage, and fat cells. As stem cells, they are an exception because they act pluripotently and can specialize in the cells of any germ layer.
- Neural cells give rise to nerve cells and their supporting cells—oligodendrocytes and astrocytes.
- Haematopoietic stem cells form all kinds of blood cells: red, white, and platelets.
- Skin stem cells form, for example, keratinocytes, which form a protective layer of skin.
- The proliferation time of somatic stem cells is longer than that of ESCs.
- It is possible to reprogram adult stem cells back to their pluripotent state.
- This can be performed by transferring the adult nucleus into the cytoplasm of an oocyte or by fusion with the pluripotent cell. The same technique was used during cloning of the famous Dolly sheep.
- hESCs are involved in whole-body development. They can differentiate into pluripotent, totipotent, multipotent, and unipotent cells.
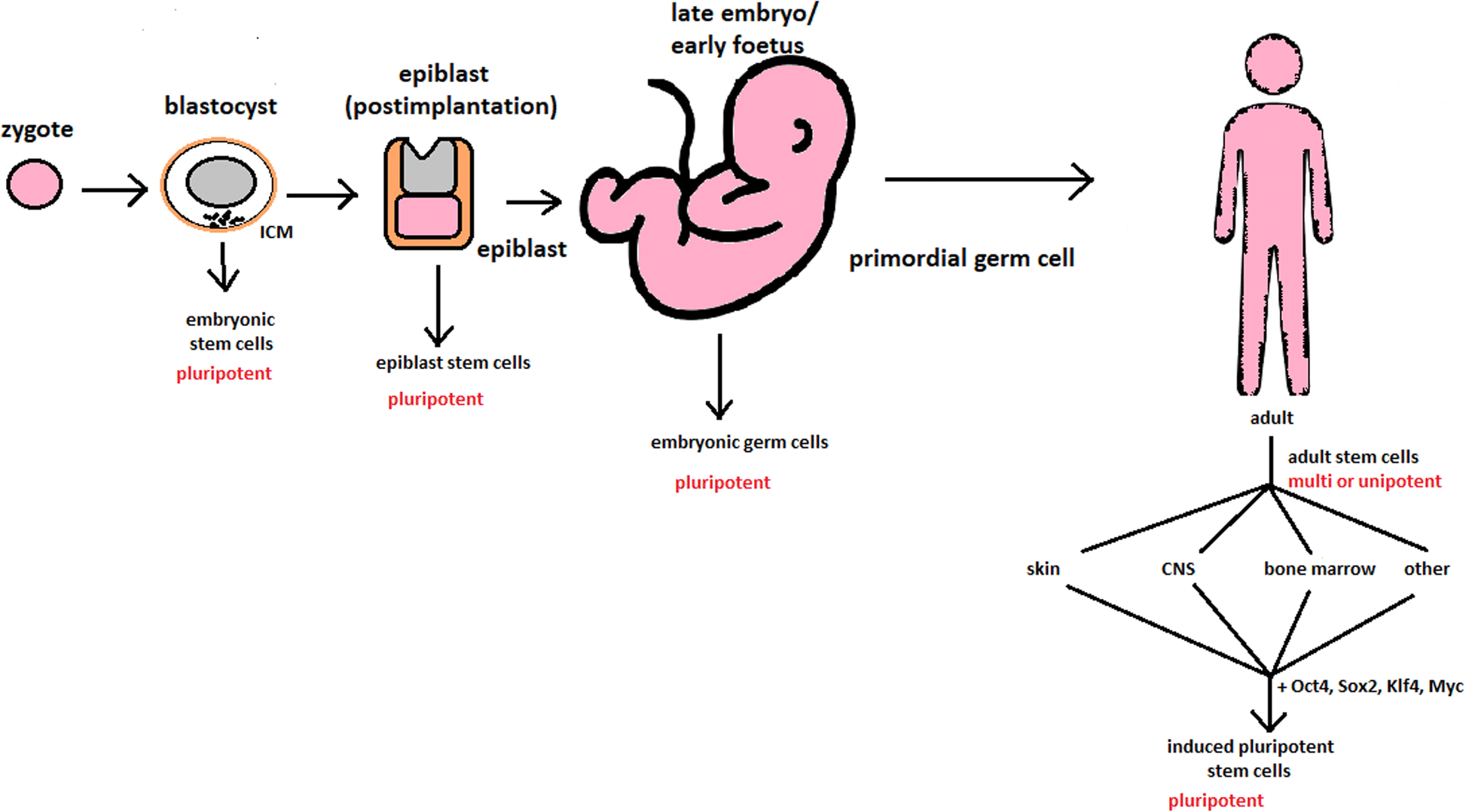
Potency ranges from pluripotent cells of the blastocyst to unipotent cells of a specific tissue in a human body such as the skin, CNS, or bone marrow. Pluripotent cells can be named totipotent if they can additionally form extraembryonic tissues of the embryo. Multipotent cells are restricted in differentiating to each cell type of given tissue. When tissue contains only one lineage of cells, stem cells that form them are called either called oligo- or unipotent.
Culturing of pluripotent stem cells in vitro. Three days after fertilization, totipotent cells are formed. Blastocysts with ICM are formed on the sixth day after fertilization. Pluripotent stem cells from ICM can then be successfully transmitted on a dish.
TURNING POINT IN STEM CELL THERAPY
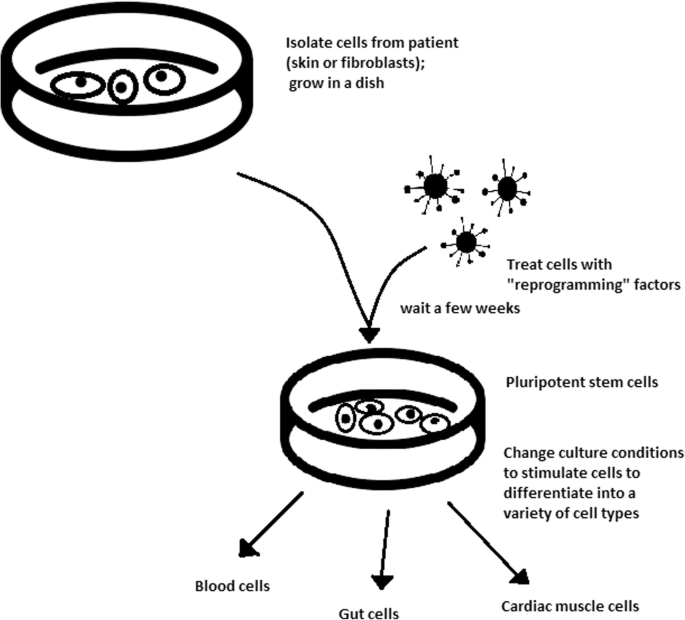
- The turning point in stem cell therapy appeared in 2006, when scientists Shinya Yamanaka, together with Kazutoshi Takahashi, discovered that it is possible to reprogram multipotent adult stem cells to the pluripotent state.
- This process avoided endangering the foetus’ life in the process.
- Retrovirus-mediated transduction of mouse fibroblasts could induce the fibroblasts to become pluripotent.
- This new form of stem cells was named iPSCs. One year later, the experiment also succeeded with human cells.
- After this success, the method opened a new field in stem cell research with a generation of iPSC lines that can be customized and biocompatible with the patient.
The turning point was influenced by former discoveries that happened in 1962 and 1987.
- The former discovery was about scientist John Gurdon successfully cloning frogs by transferring a nucleus from a frog’s somatic cells into an oocyte. This caused a complete reversion of somatic cell development. The results of his experiment became an immense discovery since it was previously believed that cell differentiation is a one-way street only, but his experiment suggested the opposite and demonstrated that it is even possible for a somatic cell to again acquire pluripotency.
- The latter was a discovery made by Davis R.L. that focused on fibroblast DNA subtraction showing that reprogramming cells is possible, and it can even be used to transform cells from one lineage to another.
iPSCs
- Although pluripotency can occur naturally only in embryonic stem cells, it is possible to induce terminally differentiated cells to become pluripotent again.
- The process of direct reprogramming converts differentiated somatic cells into iPSC lines that can form all cell types of an organism.
Source of iPSCs
- Because pluripotent cells can propagate indefinitely and differentiate into any kind of cell, they can be an unlimited source, either for replacing lost or diseased tissues.
- iPSCs bypass the need for embryos in stem cell therapy. Because they are made from the patient’s own cells, they are autologous and no longer generate any risk of immune rejection.
- At first, fibroblasts were used as a source of iPSCs. Because a biopsy was needed to achieve these types of cells, the technique underwent further research.
- Other cells were used in the process: peripheral blood cells, keratinocytes, and renal epithelial cells found in urine. An alternative strategy to stem cell transplantation can be stimulating a patient’s endogenous stem cells to divide or differentiate, occurring naturally when skin wounds are healing.
- The best stem cell source appears to be the fibroblasts, which is more tempting in the case of logistics since its stimulation can be fast and better controlled.
STEM CELL USE IN MEDICINE
- In addition to the fact that they play a large role in developing restorative medicine, their study reveals much information about the complex events that happen during human development.
- The difference between a stem cell and a differentiated cell is reflected in the cells’ DNA. In the former cell, DNA is arranged loosely with working genes. When signals enter the cell and the differentiation process begins, genes that are no longer needed are shut down, but genes required for the specialized function will remain active. This process can be reversed, and it is known that such pluripotency can be achieved by interaction in gene sequences.
- Currently, several stem cell therapies are possible, among which are treatments for spinal cord injury, heart failure, retinal and macular degeneration, tendon ruptures, and diabetes type 1.
- Multipotent haematopoietic stem cell (HSC) transplantation is currently the most popular stem cell therapy. HSC transplantation solves problems that are caused by inappropriate functioning of the haematopoietic system, which includes diseases such as leukaemia and anaemia.
- Stem cells can be used in new drug tests.
- Stem cells can be used as an alternative for arthroplasty.
- Rejuvenation by cell programming: Ageing is a reversible epigenetic process. There is a hypothesis that when human or mouse adult somatic cells are reprogrammed to iPSCs, their epigenetic age is virtually reset to zero.
- Stem cells can be induced to become a specific cell type that is required to repair damaged or destroyed tissues. Currently, when the need for transplantable tissues and organs outweighs the possible supply, stem cells appear to be a perfect solution for the problem. The most common conditions that benefit from such therapy are macular degenerations, strokes, osteoarthritis, neurodegenerative diseases, and diabetes. Due to this technique, it can become possible to generate healthy heart muscle cells and later transplant them to patients with heart disease.
- iPS cells can be stored in a tissue bank to be an essential source of human tissue used for medical examination.
- In 2011, two researchers, Katsuhiko Hayashi et al., showed in an experiment on mice that it is possible to form sperm from iPSCs.
- Thanks to stem cell therapy, it is possible not only to delay the progression of incurable neurodegenerative diseases such as Parkinson’s disease, Alzheimer’s disease (AD), and Huntington disease, but also, most importantly, to remove the source of the problem. It can also be used in dentistry.

CHALLENGES
- Although stem cells appear to be an ideal solution for medicine, there are still many obstacles that need to be overcome in the future. One of the first problems is ethical concern.
- The second problem is to achieve successful immunological tolerance between stem cells and the patient’s body.
- Additionally, there is a concern about the possibility of obtaining stem cells without the risk of morbidity or pain for either the patient or the donor.
- One of the arguments that limit the use of iPSCs is their infamous role in tumourigenicity.
STEM CELLS OBSTACLES IN THE FUTURE
- First, the most important one is about fully understanding the mechanism by which stem cells function first in animal models.
- The efficiency of stem cell-directed differentiation must be improved to make stem cells more reliable and trustworthy for a regular patient.
- The scale of the procedure is another challenge.
- The identification and proper isolation of stem cells from a patient’s tissues is another challenge.
- Immunological rejection is a major barrier to successful stem cell transplantation.
- One of the ideas that can make stem cells a “failsafe” is about implementing a self-destruct option if they become dangerous.
After several decades of experiments, stem cell therapy is becoming a magnificent game changer for medicine. With each experiment, the capabilities of stem cells are growing, although there are still many obstacles to overcome. Regardless, the influence of stem cells in regenerative medicine and transplantology is immense. Currently, untreatable neurodegenerative diseases have the possibility of becoming treatable with stem cell therapy. Induced pluripotency enables the use of a patient’s own cells. Tissue banks are becoming increasingly popular, as they gather cells that are the source of regenerative medicine in a struggle against present and future diseases. With stem cell therapy and all its regenerative benefits, we are better able to prolong human life than at any time in history.









